At Amplion, we see biomarkers as the key to delivering on the promise of personalized medicine. From drug research and discovery and companion diagnostic development to patient care, we understand that biomarkers will drive personalized care.
 Health Decisions CRO+ reports that precision medicine is making surprisingly rapid progress in drug development: In 2015, 28% of novel new drugs (NNDs) approved by FDA were precision (personalized) medicines, up from 21% in 2014.
Health Decisions CRO+ reports that precision medicine is making surprisingly rapid progress in drug development: In 2015, 28% of novel new drugs (NNDs) approved by FDA were precision (personalized) medicines, up from 21% in 2014.
In Health Decisions’ recent white paper, Nine Tips for Success in Precision Medicine: Concise Guidance for Biopharma and Diagnostics Developers, they outline the challenging, but bright, and fast approaching transition to precision medicine and states that a focus early on clinical validation of biomarkers is one of nine key steps to secure a position in the development of precision medicine.
Click here to download the Health Decisions white paper.
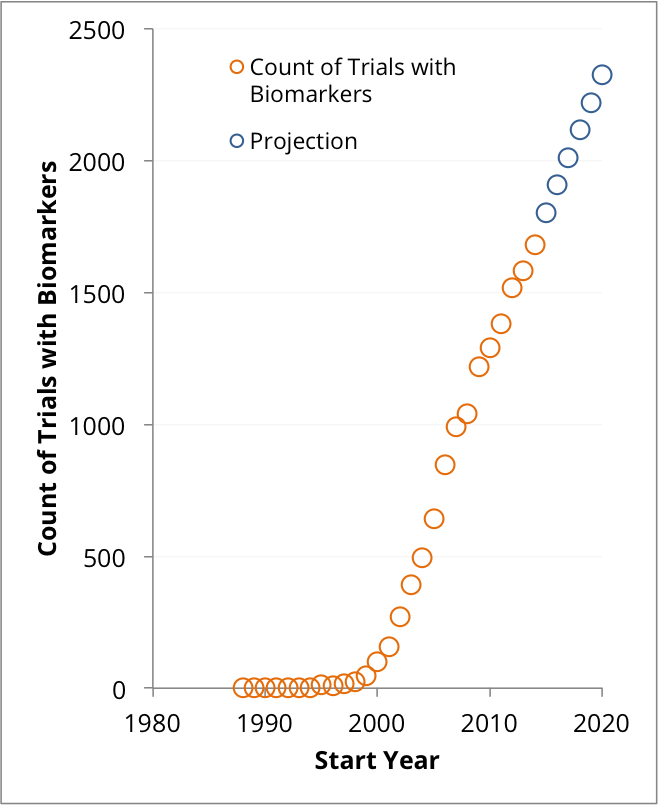
The white paper referenced our analysis of the number of clinical trials started each year in which biomarkers are being measured, and how there is a correlation between the number of unique biomarkers that a company used and the number of drug approvals that it received in 2015.