
The total number of novel biomarkers that have come to market in new IVD tests cleared or approved by the FDA over the last 12 years has been very low; an average of just 3 per year*. Is this cause for alarm? At first glance one would assume yes, but the next factor to understand is how long it takes for a biomarker to make the journey from initial discovery to a clinical test.
A long average journey means that we are still far from beginning to reap the fruits of the Human Genome Project (HGP), and the massive increase in new biomarker discoveries that it has yielded since it was completed in 2003.
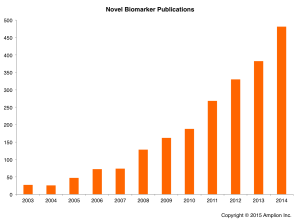 This chart shows the number of publications annually that include the words “novel biomarker” or “new biomarker” in the title or abstract. While these publications represent the most frank examples of new biomarker discoveries, they are also but a subset of the total number of new biomarkers described in the literature each year.
This chart shows the number of publications annually that include the words “novel biomarker” or “new biomarker” in the title or abstract. While these publications represent the most frank examples of new biomarker discoveries, they are also but a subset of the total number of new biomarkers described in the literature each year.To understand the average length of the biomarker validation journey we first created a list from BiomarkerBase of every novel biomarker introduced in the last 12 years (excluding those in the Mammaprint® and Prosigna® tests, and also two companion diagnostic biomarkers, CYP2C19 and UGT1A1, which are not specific to a single disease), a biomarker by our definition being the combination of a single gene or other molecular measurand, and a disease. We then identified the year during which each biomarker first appeared in a new test that was cleared or approved by the FDA.
Using this list we searched Pubmed to find the first published work that mentions each biomarker on the list, and calculated the number of years between that publication date and the test release date.
As the title of this posts foretells, the mean period from discovery to test release was 15 years across 37 different biomarkers. The standard deviation was 7.8 years, so there is quite a bit of variability in the data set.
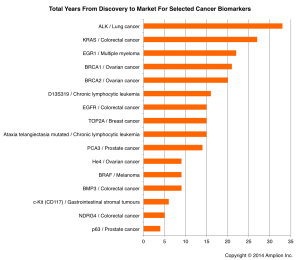 This chart shows the total number of years between discovery and first test release for 16 cancer biomarkers.
This chart shows the total number of years between discovery and first test release for 16 cancer biomarkers.Looking at just the 16 novel cancer biomarkers released in the last 12 years, the average is exactly the same as for all novel biomarkers.
The median year in which each biomarker was first published was 1993, so if we are looking to evaluate the yield of new clinical biomarkers from the HGP, and if average development periods are remaining consistent, then we are still some years away from being able to reasonably expect a huge boom in new clinical biomarkers.
*Excluding the Mammaprint® and Prosigna® tests, which are huge outliers in the landscape of FDA-reviewed tests, as they are tests that measure dozens of biomarkers in parallel, many of which are novel.
