Here at Amplion we are currently thinking very hard about how to represent in BiomarkerBase the ways in which emerging biomarkers are naturally grouping into panels, based on how they are used in clinical trials, described in the literature, and appear in clinical tests.
In this context I thought it would be interesting, as a first pass, to take a look at all of the biomarker panels that are used in FDA-cleared and approved tests* (information that is readily available in BiomarkerBase) to get a sense of how many such panels there are, in what disease areas they are used, and what the annual release rates have been over the last decade or so.
The results of this analysis have indeed proven interesting, if unsurprising, in that they reveal how few truly novel biomarker panels have come to market through FDA routes.
*In this post, and indeed throughout this blog, the terms “cleared and approved” and “approved” are used to mean all tests that have been 1) cleared by FDA using the 510(k) program, or 2) approved by FDA using the PMA program, unless specific reference is made to only PMA-approved tests.
Annual Approvals of Multi-Biomarker Tests
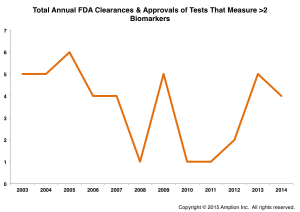
Since 2003 FDA has cleared or approved a total of 139 tests that measure 2 or more biomarkers (which I suppose is the broadest possible definition of a biomarker “panel”), out of ~1,000 total biomarker-based tests cleared or approved during that period, so multi-analyte tests overall aren’t particularly rare, and the mean number of targets per test across all tests (1.4) bears this out.
Applying a slightly more stringent definition to the concept of a “panel,” by focusing only on tests that measure three or more targets, and filtering approvals of tests that are simply updates to previously-approved tests, we quickly reduce the number of unique approvals to 43 since 2003. The average number of clearances and approvals per year is 3.6 over the period.
We have previously reported that an average of only three novel biomarker targets have been cleared or approved each year by FDA over the last decade, and if one measures test innovation in terms of the novelty of content and overall test complexity, FDA-regulated tests are clearly not where innovation is taking place in the testing market.
Panel Compositions and Uses
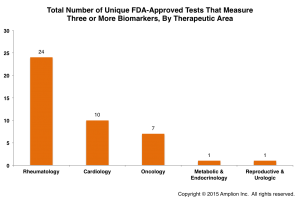
Of the 43 unique multi-biomarker tests, almost half (19) are panels for measuring anti-nuclear antibodies (ANA tests), which are used to aid in diagnosing autoimmune disorders such as lupus and scleroderma. These are surely useful tests for a lot of patients worldwide, but, since all of the panels tend to include the same markers, also represent a lot of “me-too” activity on the part of test developers.
Of the 24 non-ANA multi-biomarker tests, almost half again (10) are for measuring common cardiac markers (typically some combination of CKMB, myoglobin, troponin I, and d-dimer). The remaining 14 panels are used for a variety of diseases, with cancer tests accounting for half, and additional autoimmune disorders such as vasculitis, thrombophilia, and autoimmune cirrhosis accounting for the rest.
When grouped by therapeutic area, Rheumatology dominates.
True Multiplexing Versus Bundled Tests
An important distinction should be made between true multiplexing tests, where multiple biomarkers are measured in the same sample (ideally with quantitative results being interpreted by an algorithm), versus tests that are simply composed of multiple independent assays bundled together.
Most of the tests described in this post fall into the latter category, as there are only a handful (
It should be noted that there are examples of tests that use an algorithm to analyze the results of multiple parallel assays (the Cologuard® test from Exact Sciences being one example), and also tests that measure multiple biomarkers simultaneously, but which are not interpreted using an algorithm (the BioPlex® Vasculitis test from Bio-Rad being one example).
The most sophisticated tests, I would argue, are those that do both. They measure multiple biomarkers simultaneously, and thus utilize the smallest amount of sample material, and they use an algorithm to provide the most accurate interpretation of (ideally quantitative) measurements.
The best FDA-approved examples of such tests are the MammaPrint® test from Agendia, and the recently-released Prosigna® test from Nanostring.
Just two leading-edge biomarker panels released through FDA review in twelve years is hardly something to get excited about, but FDA has recently promised that it will adapt its review processes to a rapidly changing technical world.
Regulatory Challenges
FDA has stated that it intends to rely more on databases for the clinical validation of complex tests, in order to speed clinical validation of new markers. Margaret Hamburg, soon-to-be-former head of the Agency, recently pointed to the approval of an Illumina test for cystic fibrosis as an example of such an approach.
The review and approval of the Illumina test utilized a database provided by Johns Hopkins to validate clinically relevant variations of the CFTR gene. While this is a meaningful first step, it was limited to validating variants of a single biomarker, and it remains to be seen if such an approach will extend to multi-biomarker tests.
Until FDA adopts an approach to validation that is more dynamic, and able to deal efficiently with tests that use multiple biomarkers and variants thereof, and which can allow for rapid review and approval of inevitable and necessary updates to such tests, I fear that we will still be left with scant few examples of true precision medicine coming through FDA-reviewed channels.
We are only now starting to pull information about lab-developed tests (LDTs) into BiomarkerBase, and it will be interesting to see if there has been more innovation in this segment of the market. Anecdotal evidence would suggest that there has been, and further supports trepidation over FDA’s proposal to regulate LDTs.