In a follow on to our trial filtering changes we made in February, this week we have added a couple more options to trial filtering:
- Filtering biomarkers to those used as outcome and safety measures
- Filtering trials to only those with results
- Insights view of biomarker targets showing company and therapeutic area activity
Filtering biomarkers by outcome and safety measures
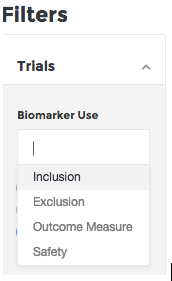
If you are planning a trial or looking for business development opportunities you can now look for specific usages of a biomarker that best fits your needs. Just log in to BiomarkerBase, browse to the Biomarkers view and select the Biomarker Use drop down. You’ll see two new options: Outcome Measure and Safety.
Filtering trials to only those with results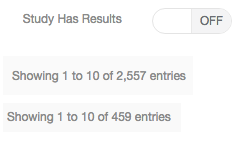
Are you trying to determine if the use of a biomarker contributed to the success of a trial or not? By indicating which trials have published results we get you much closer to that answer by allowing you to filter the trials of a biomarker to those with published results. We then still provide the simple and efficient access to the details of each trial.
Biomarker Target Insights
BiomarkerBase users tell us that a key use case for BiomarkerBase is understanding the commercial landscape. While BiomarkerBase does a good job of illustrating the commercial activity of a biomarker through our various views we can make it better. We’ve taken the first steps in improving commercial activity by adding an Insights tab to our Target view showing the companies involved in a biomarker and how they are involved (Drugs, Tests, Trials) and the Therapeutic Areas the biomarker is influencing:
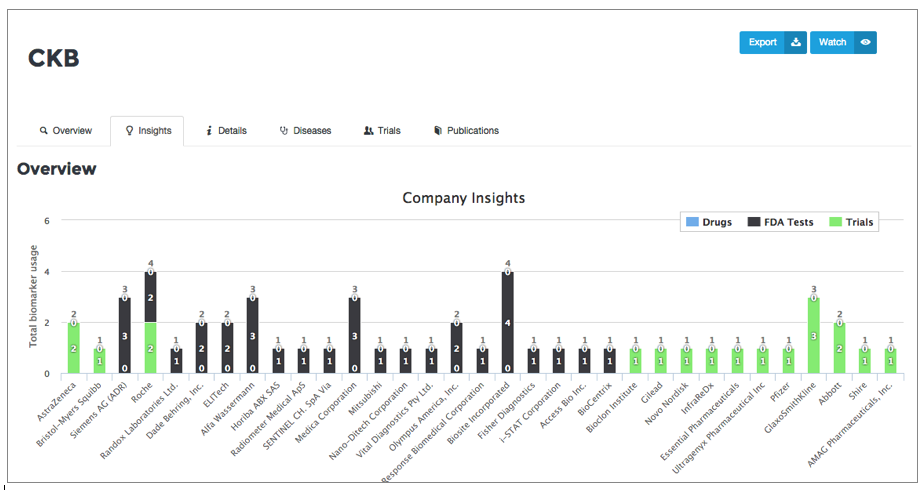
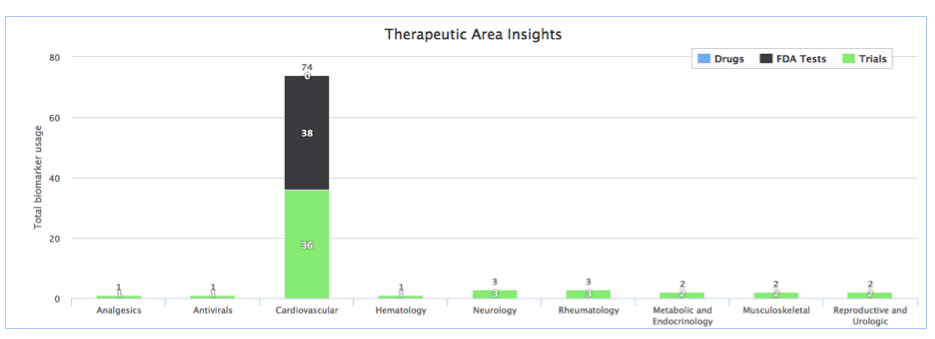
Stay tuned for what’s coming next as we continue to focus on improving our clinical trial data coverage and functionality. We are actively working to expand coverage to include EU Trials and broaden inclusion of more LDTs in the coming months.
Would you like to find out more about BiomarkerBase? Request a demo or read Health Decision’s recent white paper about how an early focus on clinical validation of biomarkers is one of nine steps to secure a position in the development of precision medicine:
[plsc_button url=”https://amplion.wpengine.com/biomarkerbase-demo/?hsCtaTracking=f9d217c0-3d3d-4625-adcc-7be5c13cb9be%7C379a477c-1ce0-462d-87d0-ff5cadcfa342″ target=”_blank” color=”green” style=”flat” radius=”square” size=”lg”]REQUEST DEMO[/plsc_button] [plsc_button url=”https://amplion.wpengine.com/download-the-health-decisions-white-paper?hsCtaTracking=0807ed16-1b6e-48df-aa06-6e0810e863c3%7Cb6c7d555-bc6b-4a83-a5e3-f9af9580483d” target=”_blank” color=”green” style=”flat” radius=”square” size=”lg”]WHITE PAPER[/plsc_button]
