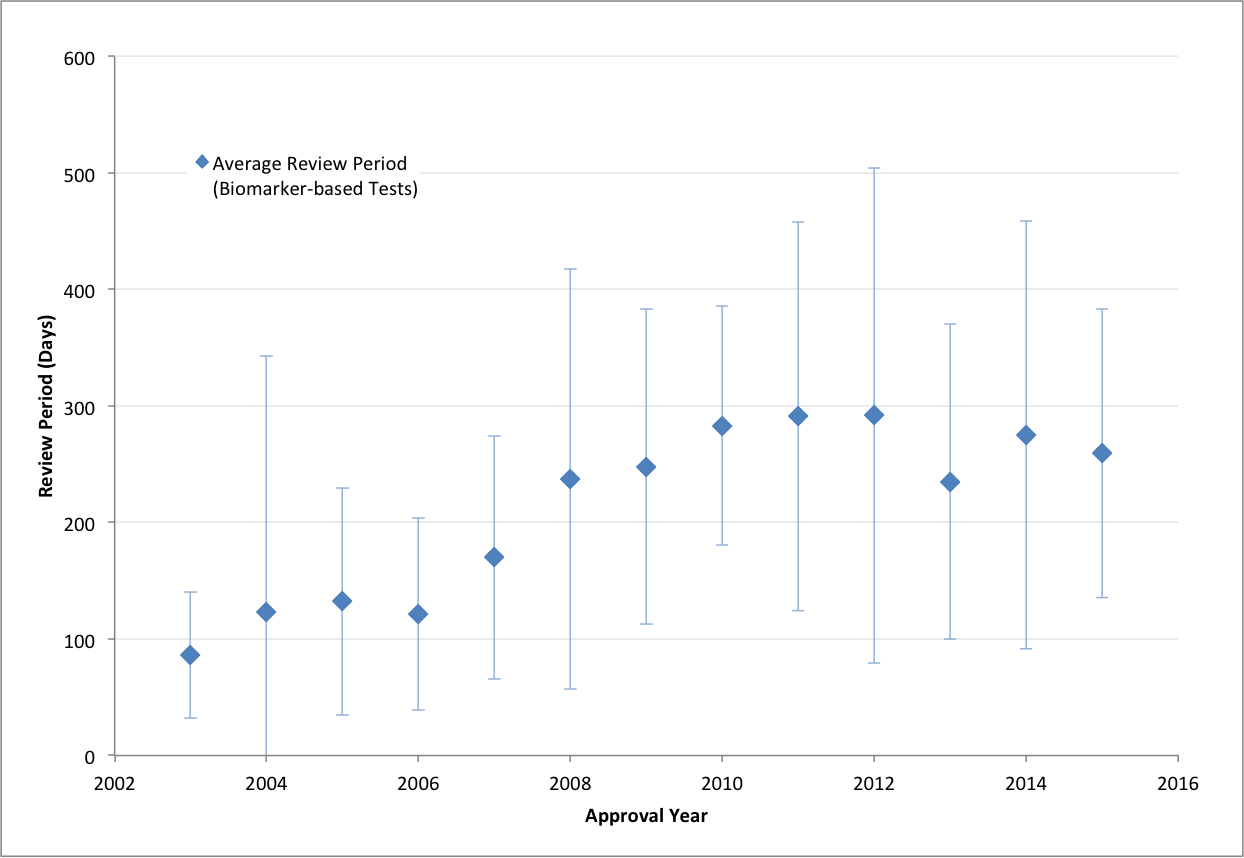
The FDA review process has a major impact on the time-to-market for tests and, therefore, on diagnostic companies’ willingness to invest in developing innovative tests for submission. The development of innovative diagnostic tests is critical to delivering on the promise of personalized medicine, which is what Amplion is all about.
Good news: The previously increasing average review period is flattening out and even decreasing.
Share this Image On Your Site
Please include attribution to www.amplion.com with this graphic.