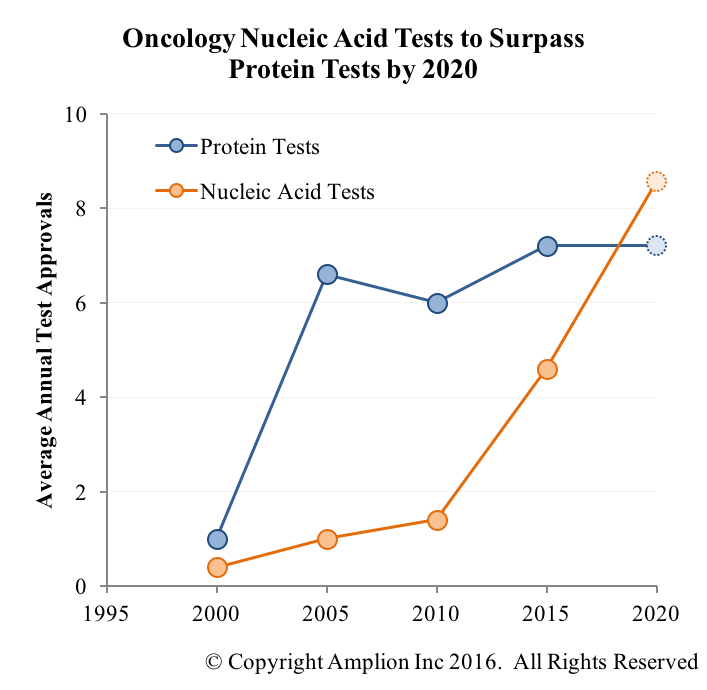
This week we took a look at FDA-cleared and approved IVDs for oncology, and grouped them based on whether they measure proteins or nucleic acids. Protein-based tests have historically been the dominant type used in the clinic, but this is changing rapidly. Based on our just-completed forecast, nucleic acid-based test approvals will surpass protein-based tests by 2020.
Tests that measure nucleic acids are generally cheaper and easier to make than those that measure proteins, especially when a company has a well-developed PCR, ISH, or sequencing platform. Such platforms are also generally more amenable to the development of multi-analyte tests.
The one exception in the protein world is testing with mass spec. Mass spec has always been very sensitive and specific, but adoption in the clinic has historically been hindered by high cost and complexity. Recent advancements have brought these factors down, however, and are behind the increasing use of mass spec in the clinic. Since there will always be a need to measure protein biomarkers it will be interesting to see if mass spec ever overtakes the immunoassay as the dominant protein measurement technology.
An interesting follow-up analysis will be to look at growth rates for mass spec assays in FDA tests and LDTs to see if there is significant recent growth.
Share this Image On Your Site
Please include attribution to amplion.com with this graphic.
Want help applying similar analysis to your projects:
[plsc_button url=”https://amplion.wpengine.com/consulting?hsCtaTracking=0ecabe42-acda-48e9-b2bd-1a020165bdb3%7Cb6240f02-690a-44c9-8446-a7209594c70a” target=”_blank” color=”green” style=”flat” radius=”square” size=”lg”]TALK TO A CONSULTANT[/plsc_button]