For the first time we will be including clinical trial data in our updated research report, Commercial Trends for Biomarker-Based IVD Tests, and here is an early look at some data describing how biomarkers are being used in drug development.
Biomarker Use by Trial Phase
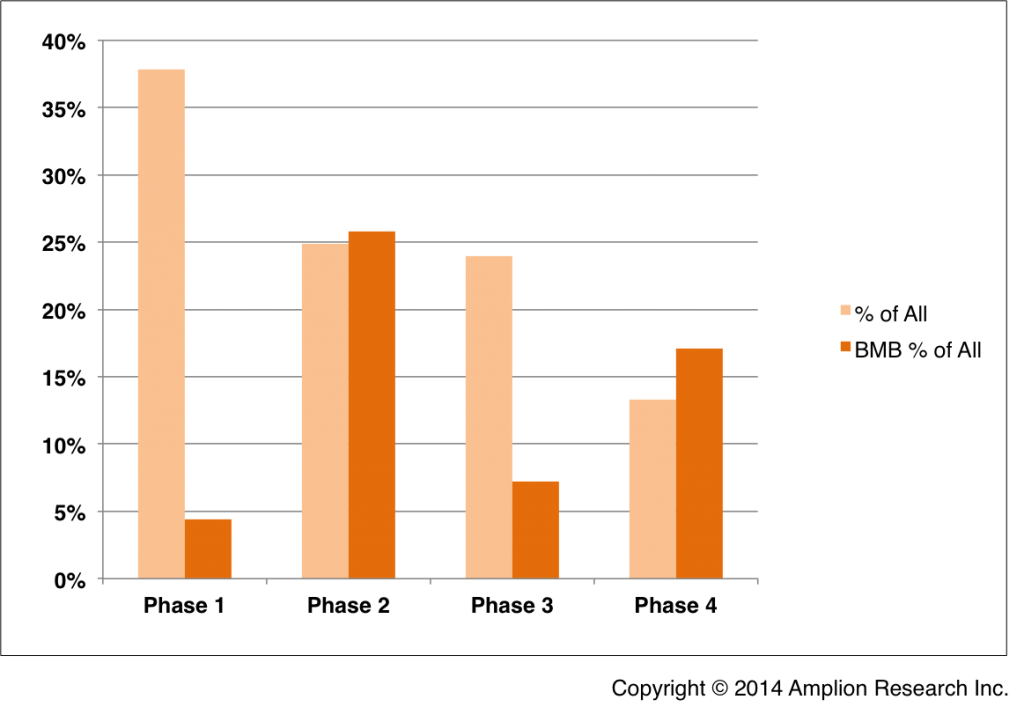
For clinical trials registered in 2013, biomarkers are being used most often in phase 2 trials.
Biomarkers are listed in approximately 25% of all phase 2 clinical trials registered during 2013, more than double the average across all trial phases.
In all, approximately 12% of all clinical trials registered during 2013 include biomarkers in the Outcome Measures and/or the Inclusion Criteria.
This figure shows the percentage of all trials represented by those in each phase (light orange), and the percentage of those in each phase that include disclosed biomarkers in the Outcome Measures or Inclusion Criteria (dark orange). Trials with biomarkers are labeled as “BMB Trials,” due to their inclusion in BiomarkerBase™. Unphased trials are not included, nor are phase 2/3 trials (The highest use of biomarkers in phase 2 makes sense when considering where in the clinical development process biomarkers are most likely to be evaluated in terms of utility for patient segmentation, as efficacy endpoints, etc.
As I was reminded during discussions with drug development professionals at the Biomarker Summit last week, phase 1 trials utilize study populations that are too small for biomarker development, and phase 3 trials include mostly well-developed biomarkers, both because of testing costs and because of the very short timelines available to plan a phase 3 study.
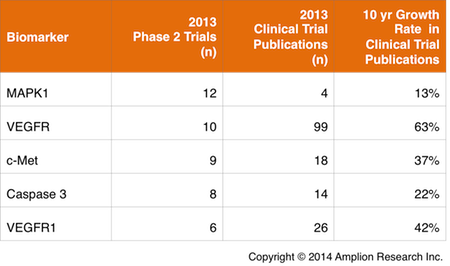
This is further supported by the diversity of targets that are measured in phase 2. Of the at least 400 biomarkers listed in 2013 trial records, over half are listed only in phase 2 trials.
Five of the top “phase 2 only” biomarkers are listed in the table below. The clinical publication track record for each biomarker is also listed, and it’s clear that for this set at least, the extent of each biomarker’s representation in 2013 clinical trials would be difficult to predict based on publication history.
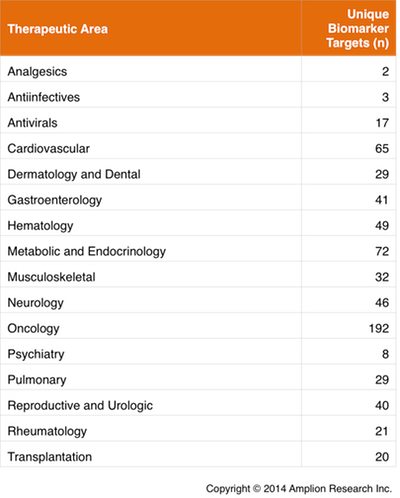
Biomarker Use by Therapeutic Area
Oncology continues to be the therapeutic area within which significantly the largest number of unique targets is being measured. Almost half of the unique biomarker targets that were listed in 2013 clinical trial records are being measured in oncology trials.
In a later post we will look at the use of biomarkers across therapeutic areas. In the current analysis we were able to determine that each unique biomarker target is used in an average of 2 different therapeutic areas, so it will be interesting to see where the biggest overlaps occur.